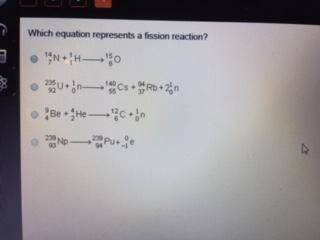
Saturated is at equilibrium so rates dissolving equals rates crystallizingĤ6 Which formula represents the product of the addition reaction between ethene and chlorine Cl 2?Ĥ7 Based on Reference Table J. 
(4) The concentration of the solid is greater than the concentration of the solution. (3) The concentration of the solid is equal to the concentration of the solution. (2) The rate of disso lving is greater than the rate of crystallization. (1) The rate of disso lvin g is equal to the rate of c rys tallizaion, Heat of reaction=products (C)- reactants (A+B)Ĥ5 Some solid KNO 3 remains at the bottom of a stoppered flask containing a saturated KNO 3(aq) solution at 22☌, Which statement explains why the contents of the fl ask are at equilibrium? If each interval on the axis labeled "Potential Energy (kJ/mol)" represents 10. Superscript 235 subscript 92 upper U plus superscript 1 subscript 0 n right arrow superscript 140 subscript 55 upper C s plus superscript 94 subscript 37 upper R b plus 2 superscript 1 subscript 0 n. Which half-reaction represents the reduction that occurs?Ĥ4 Given the equation and potential energy diagram representing a reaction: Correct answers: 2 question: Which equation represents a fission reaction Superscript 14 subscript 7 upper N plus superscript 1 subscript 1 upper H right arrow superscript 15 subscript 8 upper O. Questions 1-10 Questions 11-20 Questions 21-30 Questions 31-40 Questions 41-50 Questions 51-54 Questions 55-57 Questions 58-59 Questions 60-62 Questions 63-65 Questions 66-69 Questions 70-71 Questions 72-74 Questions 75-78 Questions 79-81 Questions 82-85Ĥ1 Which equation represents a change that results in an increase in disorder? Highlight to reveal answers and explanations These are examples of nuclear fusion.Regents Chemistry Exam Explanations January 2012 In each case, the two reactant nuclei give a product that has more mass than either of them. Notice how, on each side of the equation, the superscripts add up to 4 and the subscripts add up to 2. 
A typical example is the fusion of two deuterium nuclei to form a tritium nucleus and a proton: Now that we know what these symbols represent, let's see how they can be applied to a nuclear fusion. Just as in a balanced chemical equation, in a balanced nuclear equation, the sums of the superscripts and the sums of the subscripts must be equal on each side of the equation. We also use symbols for α and β particles and for protons and neutrons: Thus, the symbol for uranium-238 is ₉₂²³⁸ U (Sorry, the 92 should be directly under the 238, but I can’t do that in this editor). The Z describes the number of protons in the nucleus and determines the type of atom. The number at the lower left is the atomic number or Z. The A identifies the number of protons and neutrons in the nucleus. Using the figure to the right, write the reaction equations represented by. The number at the upper left is the mass number, also known ‘A’. Each of the elements involved in the reaction is identified by the chemical symbol. In order to write an equation for such a reaction, we must first establish some basic rules. Nuclear fusion is a process in which two or more atomic nuclei collide at a very high speed and join to form a new type of atomic nucleus that has more mass than any of the starting nuclei.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
